The XXIX National Congress of Health Informatics (Inforsalud 2026) organized by the SEIS concluded with one premise: technology has ceased to be a support and has become the driving force behind the renewal of our healthcare system. Under the chairmanship of Luciano Sáez the event brought together the leaders of the sector to define a roadmap in which the Digital Health Strategy goes beyond the public sector to fully integrate the private sector.
In this context, one of the points discussed with consensus and urgency was the definitive integration of the private electronic prescription into the healthcare circuit, with the aim of eliminating the barriers between the physician, the patient and the pharmacy and achieving the level of digitalization that the public prescription does have.
1. The 2027 horizon: “The end of paper”
1.1. The risks of paper prescriptions
The Congress, especially the General Council of Official Pharmacists’ Associations (CGCOF) analyzed why the current model of paper prescriptions with handwritten signatures in private healthcare is anachronistic and dangerous, and defined three critical pillars for its digital transition:
- Professional digital identity: mandatory use of digital certificates or qualified electronic signature that unequivocally identifies the registered prescriber.
- Electronic verification code: a unique code per prescription that prevents duplication and fraud, which is impossible to control in physical form.
- Centralized and interoperable repositories: Noemí Cívicos Villa (Director General of Digital Health) supported the thesis of Centralized Repositories. She explained that the Ministry is working to ensure that the private prescription nodes “talk” with the central public health node to guarantee patient clinical safety. As a result of this need, it is also particularly necessary for the different private prescription repositories to be connected to each other.
Why is change urgent? The Inforsalud safety table was forceful about the risks of paper versus the advantages of digital prescriptions:
- Clinical Safety: as does the system of EReceta Delonia system does, digitization allows to provide automatic alerts of interactions and incompatibilities, among other safety warnings.
- Traceability: with the digital prescription it is possible to know exactly who is prescribing, what is dispensed and when.
- Privacy: the digital prescription travels encrypted, protecting the patient’s sensitive data, which on paper is exposed to any loss.

1.2. Deadline for making digital private prescription mandatory
Although there is no immediately binding law, the private sector is looking at a horizon of 18 to 24 months (end of 2027) for paper prescriptions to disappear from pharmacies. Although Juan Fernando Muñoz did not set a date in his speech, he did insist that the new Strategic Plan 2026-2031 calls for tangible results in the first two years of the cycle. This puts the administrative pressure precisely in 2027.
It was the technical representatives of the pharmacists’ associations who pointed out that, given the current level of integration of electronic private prescriptions, paper will cease to make technical and economic sense within the two-year period for urban environments. Regarding the differences in the implementation horizon between urban and rural areas, Juan Fernando Muñoz and Noemí Cívicos mentioned that the Digital Health Strategy 2026-2031 includes specific items to eliminate the “white zones” without connectivity. In rural areas there are still areas with coverage problems or unstable connections that limit the use of systems such as private digital prescriptions.
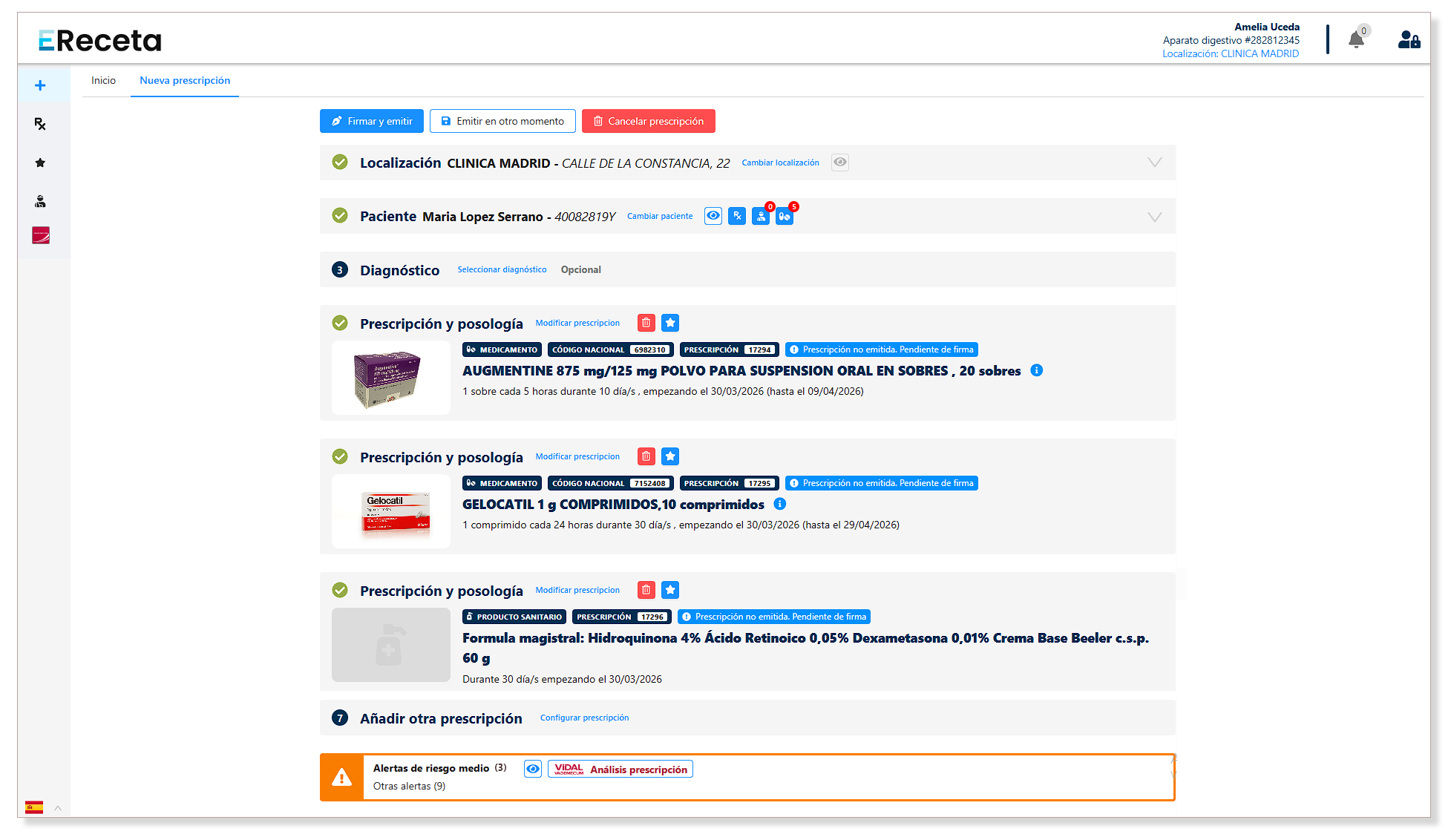
2. Delonia’s role in the digital transformation of prescriptions
In this context of homologation and technical requirements, solutions such as EReceta Delonia are positioned as a benchmark for hospitals, insurance companies and mutual insurance companies, clinical management software, independent doctors and private clinics, not only complying with CGCOM approval standards, but also providing added value:
A. EReceta Delonia has been certified in the National Security Scheme (ENS) and CE DM Class IIb and the CE DM Class IIb marking for its medical device for prescription analysis, thanks to the strategic integration with Vidal Vademécum.
B. EReceta provides key and differential functionalities:
- Advanced clinical alerts: for safer and more accurate prescribing.
- Integral management: authorized for commercial drugs, active ingredients, narcotics, psychotropic drugs, vaccines and master formulas.
- Real-time information: connection with the AEMPS to detect supply problems and to have complete information on package inserts and technical data sheets.
- Interconnectivity of dispensing with ability to provide therapeutic adherence data.
- Mutual integration: full support for MUFACE and ISFAS, not only for the digital prescription but also for visa treatments.

3. EReceta Delonia is committed to full integration in the HIS
At Delonia we advocate the full integration of EReceta in clinical management systems(HIS). This vision is strategically aligned with the Inforsalud 2026 consensus, where parallel systems and information silos were identified as the main enemies of interoperability. In order for the digital prescription to be an invisible tool for the healthcare provider, it is essential to ensure integration:
- Centralize history and prescription in a single interface, saving critical time.
- Eliminate manual entry and “copy and paste“, drastically reducing human error.
- Generate a simple workflow to facilitate physician adoption of the system.
Digital transformation implies eliminating paper. At EReceta Delonia, we lead this process with an advanced tool and continuous evolution, always meeting the needs of those who use our service.






